Nous capitalisons sur notre expertise singulière du sang tout au long de la chaîne du soin pour contribuer à la recherche scientifique, innover et développer des procédés et traitements médicaux qui bénéficieront au plus vite aux patients. Partout en France, nos équipes de chercheurs mettent leur savoir-faire au service de la science et de la médecine pour faire reculer la maladie.
Une expertise du sang appliquée à la recherche
Nous avons la responsabilité des réserves nationales de produits sanguins, de leur collecte chez des donneurs de sang bénévoles jusqu’à leur délivrance et distribution aux établissements de santé. Ces produits sanguins permettent de soigner des patients victimes d’accidents graves, atteints de maladies chroniques, de maladies génétiques ou de cancers. L’objectif est de mettre à disposition le bon produit sanguin, au bon moment, au bon patient ainsi que d'assurer le conseil transfusionnel associé si besoin.
Afin d’être toujours plus performant dans notre activité cœur de métier, la transfusion sanguine, notre établissement a structuré au fil du temps un véritable écosystème de recherche et d’innovation qui s’inscrit tout au long de la chaîne de soin. Nous cherchons à améliorer nos connaissances de la cellule, du diagnostic et la compréhension de certains phénomènes.
C’est en grande partie grâce à la valorisation des dons de sang non utilisables pour la transfusion que nous faisons de la recherche scientifique.
Notre activité porte sur :
- l’amélioration des produits sanguins que nous délivrons ;
- la prise en charge et la sécurité des donneurs de sang ;
- le soin des patients, en développant des traitements de pointe pour faire reculer les maladies.
L’un des principaux enjeux pour notre établissement est de concrétiser les projets de recherche afin que les progrès scientifiques bénéficient aux patients en améliorant leur prise en charge. Pour cela, une équipe dédiée accompagne et protège nos inventions de leur émergence jusqu’à leur promotion et leur transfert auprès de partenaires industriels afin de les rendre accessibles au plus grand nombre.
La recherche en chiffres
180
18
Une collaboration avec les plus grands acteurs de la recherche
En France comme à l’international, nos équipes collaborent, pour certains projets, avec les plus grands acteurs publics de la recherche, comme les universités, les CHU, l’Inserm ou encore l’Institut Pasteur, et des petites et moyennes entreprises ainsi que des grands groupes internationaux afin d’accéder à des technologies innovantes notamment.
Je suis en quête permanente.
Dès que je découvre un élément qui fait avancer mes recherches, je me concentre sur la suite. Il faut sans cesse se renouveler et innover pour réussir les challenges scientifiques que l’on se fixe. Avec un objectif qui reste toujours le même : assurer la sécurité des patients.
Daisy Bougard-Rossignol
Maître de recherche à l'EFS
La recherche au service des donneurs de sang
Nous développons différents projets au service de l’amélioration du don de sang et de la prise en charge des donneurs. Nos équipes s’intéressent par exemple à la sociologie du don : comprendre les motivations et les freins au don de sang pour mieux fidéliser et recruter les donneurs ; adapter la promotion du don dans un contexte de diversité culturelle. Nous menons également différents projets de recherche contribuant à la surveillance des risques : malaises vagaux lors des dons, risques infectieux (Zika, Chikungunya, Dengue, etc.). Enfin, une partie de notre recherche est orientée autour de la découverte de nouveaux groupes sanguins et de variants.

La recherche pour améliorer les produits sanguins
Nos équipes travaillent sur différents projets en lien avec les produits sanguins afin notamment d’assurer la bonne réussite des transfusions sanguines et des greffes. Nous cherchons par exemple à optimiser les produits sanguins et à mieux comprendre les mécanismes de compatibilité entre les donneurs et les patients et ce, afin d’être toujours plus performant dans notre activité cœur de métier. Nous œuvrons aussi à inventer les produits sanguins de demain en reproduisant, en laboratoire, des plaquettes ou encore les globules rouges dits de culture. Des projets ambitieux qui pourraient permettre de contribuer à accroître les réserves en produits sanguins.
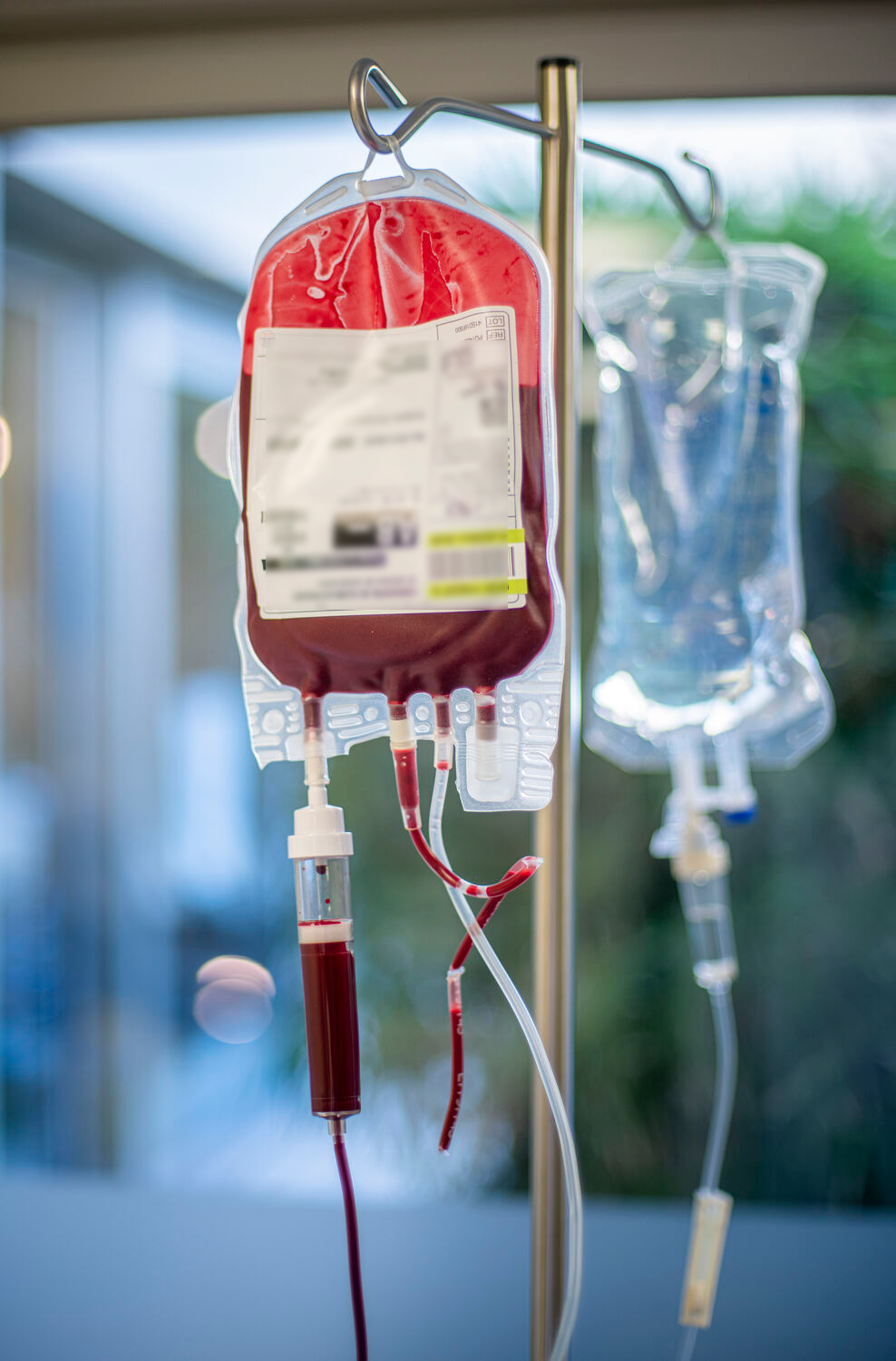
La recherche au service des patients
Une grande partie des patients transfusés ou greffés sont atteints de maladies chroniques, génétiques ou de certains cancers. C’est donc naturellement que nos équipes ont orienté une partie de leur recherche afin de faire avancer la science et reculer ces maladies. Dotées d’une expertise aiguisée de la cellule, nos équipes développent différents projets pour trouver des traitements ou mieux comprendre les maladies rares hématologiques (drépanocytose, thalassémie, etc.), les cancers du sang (leucémies, lymphomes, etc.) ou encore les maladies cardio-vasculaires comme la thrombose, les anémies. Nous contribuons alors aux évolutions médicales, notamment grâce à la médecine personnalisée qui permet une prise en charge adaptée aux spécificités biologiques de chaque patient.


